Group leader: Annalisa Buffo
Our research focuses on the role of glia and progenitor cells in brain plasticity and repair , and on the implementation of cell replacement approaches and/or training protocols to promote functional recovery in CNS diseases.
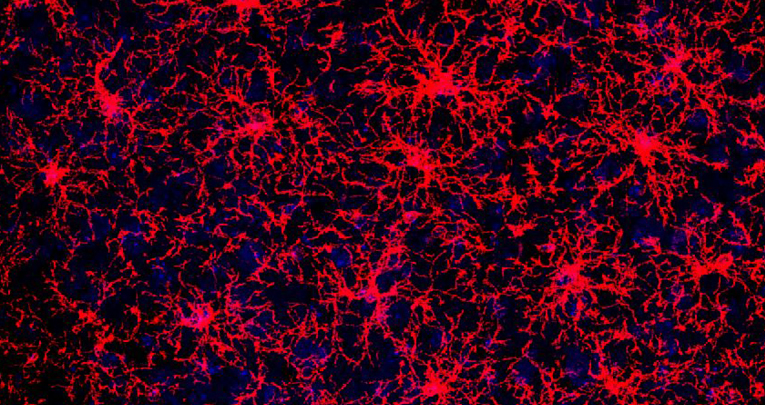
We believe that specific issues regarding glia and neural progenitors are particularly promising to both unveil new keys to the understanding of physiology, and accomplish therapeutic actions to cure disease and promote repair in acute and chronic neurodegenerative pathologies. As for astrocytes, very little is known on how distinct astroglial subtypes are specified and how much it impacts the shaping of the circuits. Further, mechanisms underlying the acquisition of stem cell properties in parenchymal astroglia remain undefined. We address these issues by studying the specification of astroglial subtypes in the cerebellum, and the latent stem cell properties of adult striatal astroglia.
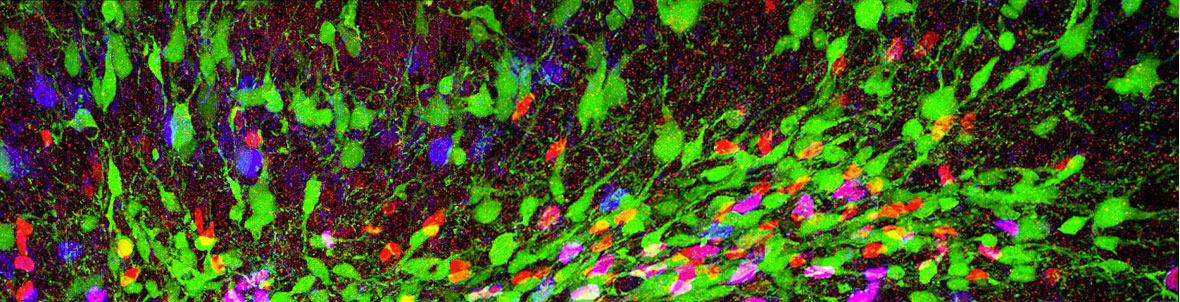
Oligodendrocyte progenitors self-maintain but have limited capability to repair myelin. Understanding their biology will help fostering myelin regeneration and reveal unsuspected functions of these progenitors in their crosstalk with neurons.
Recent advancements in human stem cell technology and reprogramming prompt the need of developing strategies to obtain proper differentiation into specific neuronal identities and functional integration in the recipient brain. Moreover, based on the efficacy of external stimuli and training to promote circuit plasticity, rehabilitation protocols appear as promising tools to promote adaptive remodelling of defective circuits and to enhance the integration of transplanted cells into the recipient tissue, therefore boosting functional recovery. We perform preclinical studies to define therapies for neurological diseases based on adaptive cell replacement and/or manipulation of circuit plasticity.
S&P BRAIN
Services and Products for preclinical proof of concepts
Our spinoff provides scientific expertise, animal models, equipment and facilities to pharmaceutical, biotechnology, and medical device Companies and to Research Centers for proof–of-concept or pilot in vivo studies.
News
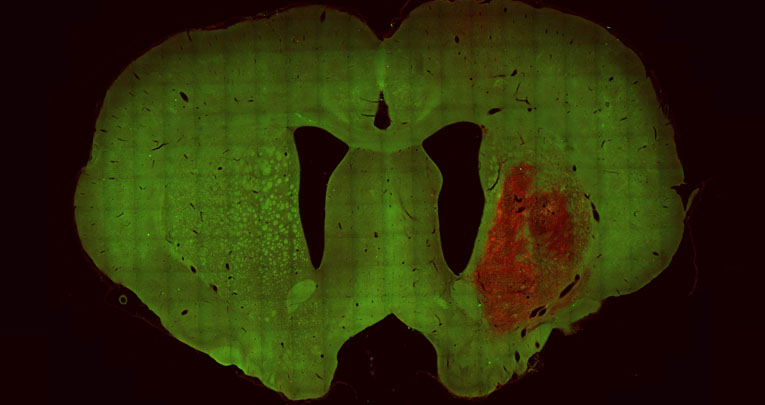
HD: human striatal progenitors transplanted in the lesioned striatum are able to survive long-term
Stem Cell Research & Therapy , 28 July 2023
Roberta Schellino, Dario Besusso, Roberta Parolisi, Gabriela B. Gómez-González, Sveva Dallere, Linda Scaramuzza, Marta Ribodino, Ilaria Campus, Paola Conforti, Malin Parmar, Marina Boido, Elena Cattaneo, Annalisa Buffo
Research Groups NICO: Physiopathology of neural stem cells and Brain development and disease
Not all oligodendrocyte precursors are created equal
Nature Communication , 28 April 2022
Molecular and functional heterogeneity in dorsal and ventral oligodendrocyte progenitor cells of the mouse forebrain in response to DNA damage.
Enrica Boda, Martina Lorenzati, Roberta Parolisi, Brian Harding, Gianmarco Pallavicini, Luca Bonfanti, Amanda Moccia, Stephanie Bielas, Ferdinando Di Cunto, Annalisa Buffo