Glia, May 2018
Sox2 conditional mutation in mouse causes ataxic symptoms, cerebellar vermis hypoplasia, and postnatal defects of Bergmann glia.
Cerrato V 1 , Mercurio S 2 , Leto K 1 , Fucà E 1 , Hoxha E 1 , Bottes S 2 , Pagin M 2 , Milanese M 3 , Ngan CY 4 , Concina G 1 , Ottolenghi S 2 , Wei CL 4 , Bonanno G 3 , Pavesi G 5 , Tempia F 1 , Buffo A 1 , Nicolis SK 2 .
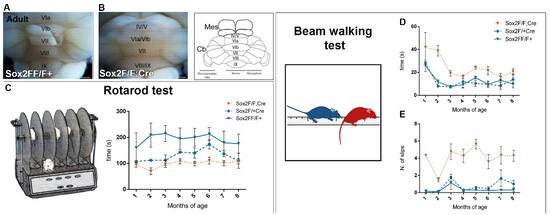
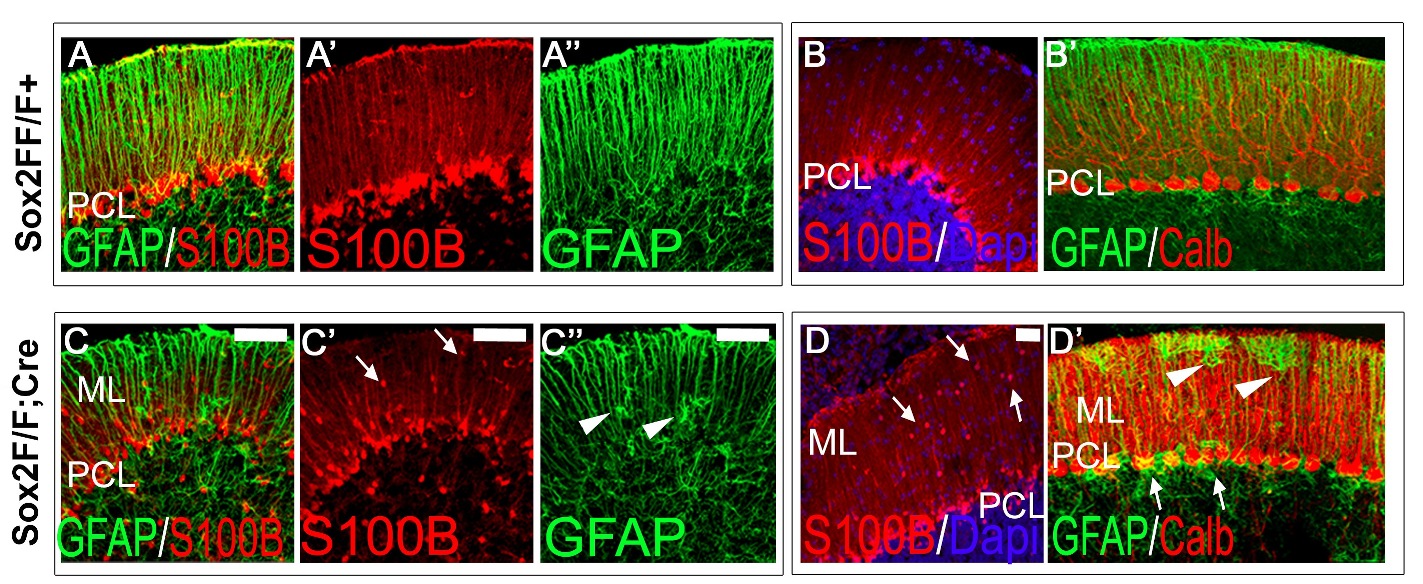
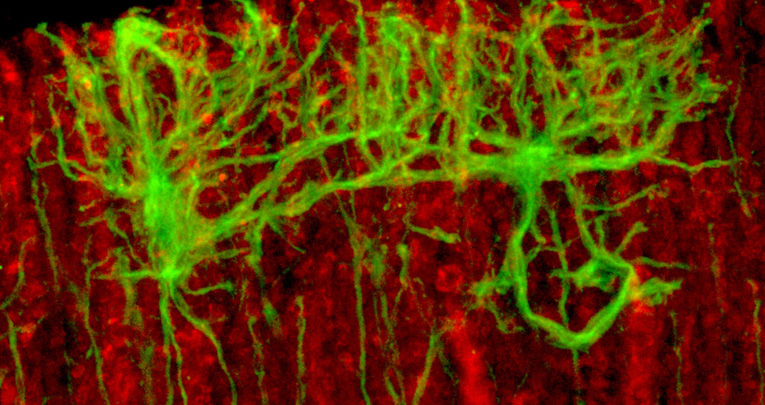
Sox2 is a transcription factor active in the nervous system, within different cell types, ranging from radial glia neural stem cells to a few specific types of differentiated glia and neurons. Mutations in the human SOX2 transcription factor gene cause various central nervous system (CNS) abnormalities, involving hippocampus and eye defects, as well as ataxia. Conditional Sox2 mutation in mouse, with different Cre transgenes, previously recapitulated different essential features of the disease, such as hippocampus and eye defects. In the cerebellum, Sox2 is active from early embryogenesis in the neural progenitors of the cerebellar primordium; Sox2 expression is maintained, postnatally, within Bergmann glia (BG), a differentiated cell type essential for Purkinje neurons functionality and correct motor control. By performing Sox2 Cre-mediated ablation in the developing and postnatal mouse cerebellum, we reproduced ataxia features. Embryonic Sox2 deletion (with Wnt1Cre) leads to reduction of the cerebellar vermis, known to be commonly related to ataxia, preceded by deregulation of Otx2 and Gbx2, critical regulators of vermis development. Postnatally, BG is progressively disorganized, mislocalized, and reduced in mutants. Sox2 postnatal deletion, specifically induced in glia (with GLAST-CreERT2), reproduces the BG defect, and causes (milder) ataxic features. Our results define a role for Sox2 in cerebellar function and development, and identify a functional requirement for Sox2 within postnatal BG, of potential relevance for ataxia in mouse mutants, and in human patients.