Journal of Neurosurgery, Jenuary 2017
Irreversible changes occurring in long-term denervated Schwann cells affect delayed nerve repair
Giulia Ronchi 1,2 , Michele Cillino 3 , Giovanna Gambarotta 1 , Benedetta Elena Fornasari 1 , Stefania Raimondo 1, 2 , Pierfrancesco Pugliese 4 , Pierluigi Tos 4 , Adriana Cordova 3 , Francesco Moschella 3 and Stefano Geuna 1, 2
Objective
Multiple factors may affect functional recovery after peripheral nerve injury, among them the lesion site and the interval between the injury and the surgical repair. When the nerve segment distal to the lesion site undergoes chronic degeneration, the ensuing regeneration (when allowed) is often poor. The aims of the current study were as follows: 1) to examine the expression changes of the neuregulin 1/ErbB system during long-term nerve degeneration; and 2) to investigate whether a chronically denervated distal nerve stump can sustain nerve regeneration of freshly axotomized axons.
Methods
This study used a rat surgical model of delayed nerve repair consisting of a cross suture between the chronically degenerated median nerve distal stump and the freshly axotomized ulnar proximal stump. Before the suture, a segment of long-term degenerated median nerve stump was harvested for analysis. Functional, morphological, morphometric, and biomolecular analyses were performed.
Results
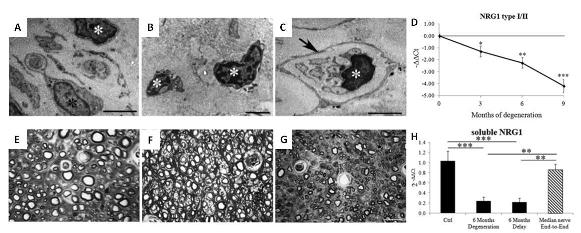
The results showed that neuregulin 1 is highly downregulated after chronic degeneration, as well as some Schwann cell markers, demonstrating that these cells undergo atrophy, which was also confirmed by ultrastructural analysis. After delayed nerve repair, it was observed that chronic degeneration of the distal nerve stump compromises nerve regeneration in terms of functional recovery, as well as the number and size of regenerated myelinated fibers. Moreover, neuregulin 1 is still downregulated after delayed regeneration.
Conclusions
The poor outcome after delayed nerve regeneration might be explained by Schwann cell impairment and the consequent ineffective support for nerve regeneration. Understanding the molecular and biological changes occurring both in the chronically degenerating nerve and in the delayed nerve repair may be useful to the development of new strategies to promote nerve regeneration. The results suggest that neuregulin 1 has an important role in Schwann cell activity after denervation, indicating that its manipulation might be a good strategy for improving outcome after delayed nerve repair.
Key Words delayed nerve repair; NRG1/ErbB system; neuregulin 1; nerve degeneration; nerve regeneration; stereology; peripheral nerve