Acta Neuropathologica , July 2019
Detrimental and protective action of microglial extracellular vesicleson myelin lesions: astrocyte involvement in remyelination failure.
Marta Lombardi 1,2 , Roberta Parolisi 3 , Federica Scaroni 2 , Elisabetta Bonfanti 4 , Alice Gualerzi 5 , Martina Gabrielli 2 , Nicole Kerlero de Rosbo 6 , Antonio Uccelli 6,7 , Paola Giussani 8 , Paola Viani 8 , Cecilia Garlanda 9 , Maria P. Abbracchio 4 , Linda Chaabane 10 , Annalisa Buffo 3 , Marta Fumagalli 4 , Claudia Verderio 2 .
Microglia are highly plastic immune cells which exist in a continuum of activation states. By shaping the function of oligodendrocyte precursor cells (OPCs), the brain cells which differentiate to myelin-forming cells, microglia participate in both myelin injury and remyelination during multiple sclerosis. However, the mode(s) of action of microglia in supporting or inhibiting myelin repair is still largely unclear.
In a recent paper published in Acta Neuropathologica , in collaboration dr. Claudia Verderio (CNR Milan), the laboratory of Pathophysiology of neural stem cells (at NICO) , supervised by prof. Annalisa Buffo (University of Turin), studied the effects of extracellular vesicles (EVs) produced in vitro by either pro-inflammatory or pro-regenerative microglia on OPCs at demyelinated lesions caused by lysolecithin injection in the mouse corpus callosum.
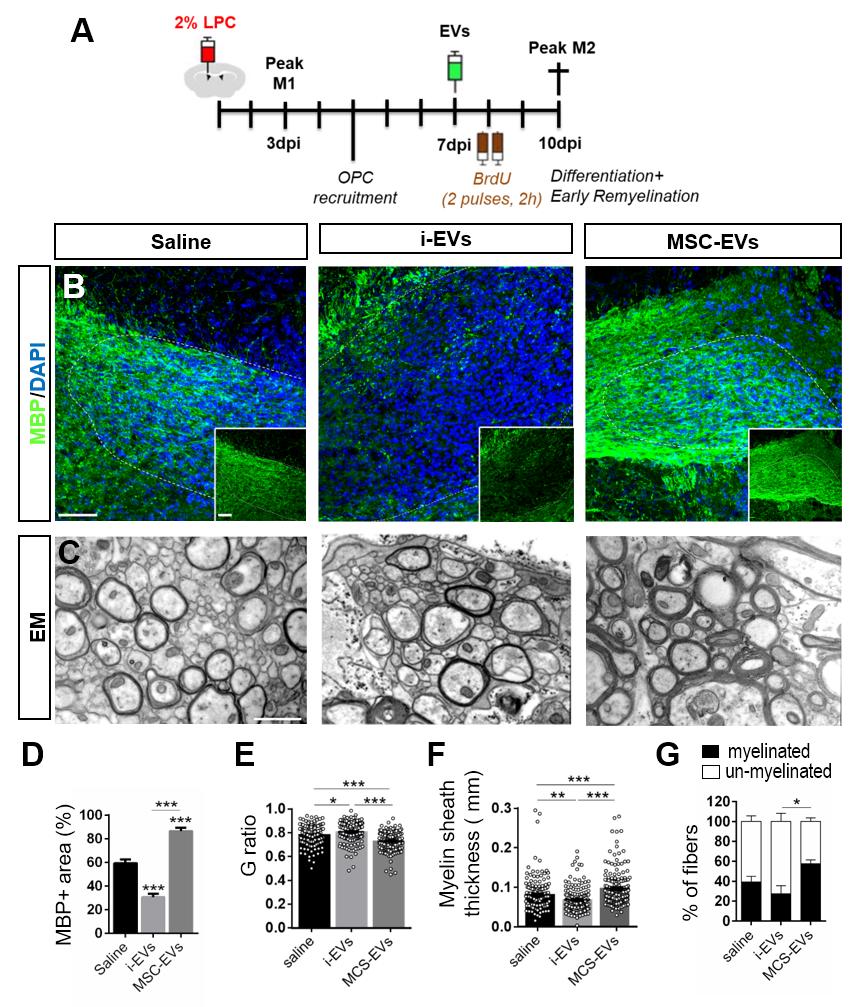
In in-vivo experiment performed by dr. Roberta Parolisi , the immunolabelling for myelin proteins and electron microscopy showed that EVs released by pro-inflammatory microglia blocked remyelination, whereas EVs produced by microglia co-cultured with immunosuppressive mesenchymal stem cells promoted OPC recruitment and myelin repair. The molecular mechanisms responsible for the harmful and beneficial EV actions were dissected in primary OPC cultures.
By exposing OPCs, cultured either aloneor with astrocytes, to inflammatory EVs, we observed a blockade of OPC maturation only in the presence of astrocytes, implicating these cells in remyelination failure. Biochemical fractionation revealed that astrocytes may be converted into harmful cells by the inflammatory EV cargo, as indicated by immunohistochemical and qPCR analyses, whereas surface lipid components of EVs promote OPC migration and/or differentiation, linking EV lipids to myelin repair. Although the mechanisms through which the lipid species enhance OPC maturation still remain to be fully defined, we provide the first demonstration that vesicular sphingosine 1 phosphate stimulates OPC migration, the first fundamental step in myelin repair.
From this study, microglial EVs emerge as multimodal and multitarget signalling mediators able to influence both OPCs and astrocytes around myelin lesions, which may be exploited to develop novel approaches for myelin repair not only in multiple sclerosis, but also in neurological and neuropsychiatric diseases characterized by demyelination.